
Spring term brought with it many new things, new classes, new racing season (rowing), and a new research project. I had been paired with Allyson through the Fisheries and Wildlife Mentorship Program to develop my research skills. A fellow undergrad, Danielle, and I would help Allyson with her mercury research.
Danielle and I started by simply educating ourselves about mercury and its effects in songbirds. Once we had a grasp on the broader implications of the effects of mercury in the environment, we turned to a question that has plagued researchers in the field: what is the best way to measure mercury in songbirds? This is a question that Allyson has been thinking about for some time, and it would become the topic for my and Danielle’s projects.
Mercury can be measured through blood, living tissue, or feather samples. Each method has pros and cons. Blood and tissue samples provide a more accurate measurement, but they are difficult to obtain. Tissue samples can only be taken from a deceased bird, therefore blood samples are most commonly used. Feathers, on the other hand are easy to obtain, but it is unclear if feathers are an accurate measurement of mercury.
We want to know if feather mercury is an accurate representation of the mercury in the bird. We are also curious if the rate of feather growth factors into mercury measurements. There would be some obvious benefits to being able to accurately measure mercury concentrations in birds using only feathers. Feathers are easier to obtain than blood, which benefits both the researchers and the birds.
Allyson had already done some work on the relationship between blood mercury and tail feather mercury. Danielle and I are exploring multiple feather tracts to determine differences in mercury concentrations between feather tracts and which tracts, if any, correlate to blood mercury levels.
Prior to our arrival in the lab, Colleen and a team of volunteers plucked and organized feathers from deceased songbirds that were donated to the lab, post-mortem, by the local wildlife rehabilitation center, Chintimini. Danielle and I both chose to focus on species in the thrush family, including American Robins, Varied Thrushes, Swanson’s Thrushes, and a Hermit Thrush.
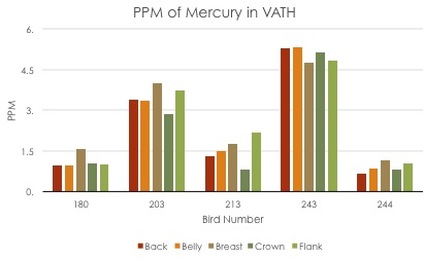
I chose to examine five body feather tracts--crown, back, breast, flank, and belly--to determine patterns in mercury levels across feather tracts. I am also looking for any patterns that arise in the mercury level across the various feather tracts.
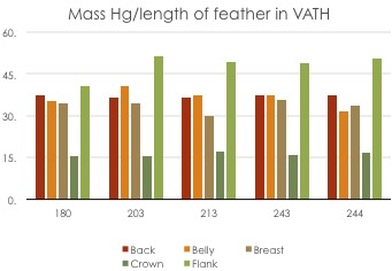
As of now, after the first round of DMA data, we still have many questions. We are excited, yet a little puzzled by our results so far. The results are variable depending on whether or not we include the rate of feather growth in our measurements (see the graphs below). Before determining any patterns in feather mercury concentration, we must first determine which measurement method is correct, or at least which one makes the most sense. After that, we will run statistical analyses to determine the significance of our results. So that is where we sit until next term.
This project has already taught me so much, from the basics of mercury, to prepping samples for the DMA analyzer, and tackling, organizing, and some analysis of unique research questions. I am excited to continue to untangle the results and to move farther in the project.
Danielle and I started by simply educating ourselves about mercury and its effects in songbirds. Once we had a grasp on the broader implications of the effects of mercury in the environment, we turned to a question that has plagued researchers in the field: what is the best way to measure mercury in songbirds? This is a question that Allyson has been thinking about for some time, and it would become the topic for my and Danielle’s projects.
Mercury can be measured through blood, living tissue, or feather samples. Each method has pros and cons. Blood and tissue samples provide a more accurate measurement, but they are difficult to obtain. Tissue samples can only be taken from a deceased bird, therefore blood samples are most commonly used. Feathers, on the other hand are easy to obtain, but it is unclear if feathers are an accurate measurement of mercury.
We want to know if feather mercury is an accurate representation of the mercury in the bird. We are also curious if the rate of feather growth factors into mercury measurements. There would be some obvious benefits to being able to accurately measure mercury concentrations in birds using only feathers. Feathers are easier to obtain than blood, which benefits both the researchers and the birds.
Allyson had already done some work on the relationship between blood mercury and tail feather mercury. Danielle and I are exploring multiple feather tracts to determine differences in mercury concentrations between feather tracts and which tracts, if any, correlate to blood mercury levels.
Prior to our arrival in the lab, Colleen and a team of volunteers plucked and organized feathers from deceased songbirds that were donated to the lab, post-mortem, by the local wildlife rehabilitation center, Chintimini. Danielle and I both chose to focus on species in the thrush family, including American Robins, Varied Thrushes, Swanson’s Thrushes, and a Hermit Thrush.
I chose to examine five body feather tracts--crown, back, breast, flank, and belly--to determine patterns in mercury levels across feather tracts. I am also looking for any patterns that arise in the mercury level across the various feather tracts.
As of now, after the first round of DMA data, we still have many questions. We are excited, yet a little puzzled by our results so far. The results are variable depending on whether or not we include the rate of feather growth in our measurements (see the graphs below). Before determining any patterns in feather mercury concentration, we must first determine which measurement method is correct, or at least which one makes the most sense. After that, we will run statistical analyses to determine the significance of our results. So that is where we sit until next term.
This project has already taught me so much, from the basics of mercury, to prepping samples for the DMA analyzer, and tackling, organizing, and some analysis of unique research questions. I am excited to continue to untangle the results and to move farther in the project.

 RSS Feed
RSS Feed
